|
1/9/2024 0 Comments Western blot transfer protocol 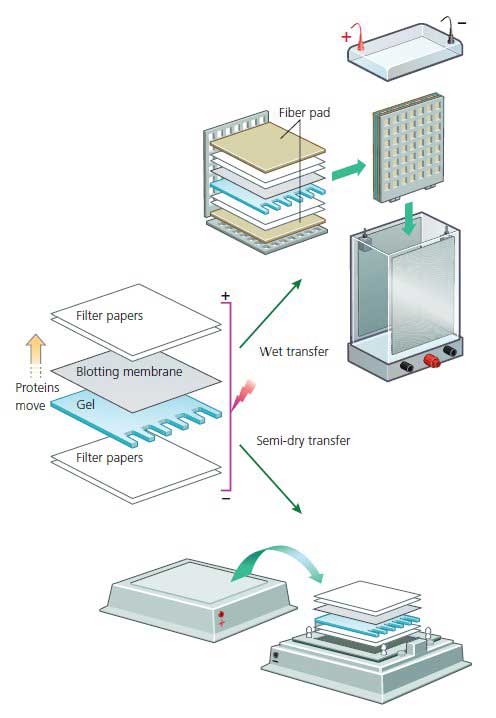
The two commonly used electro-transfer techniques are wet transfer and semi-dry transfer (Figure 1). The gel, membrane, and electrodes are assembled in a sandwich so that proteins move from gel to membrane, where they are captured, in a pattern that perfectly mirrors their migration positions in the gel. Electro-transfer relies on the same electro-mobility principles that drive the migration of proteins during separation in PAGE. Suitable for small proteins high protein binding capacity mechanical strength chemical stabilityĮlectro-transfer is almost exclusively the contemporary transfer method of choice due to its speed, uniformity of transfer, and transfer efficiency. Pre-wet in methanol before using with aqueous buffersĪmido black India ink Silver Coomassive Brilliant Blue Ponceau S Deep Purple colloidal gold The comparison of NC and PVDF membrane.Īmido black aniline blue black ponceau S Deep purple Fast green Toluidine blueįragile-limited possibilities to strip and reprobe Not recommended for small proteins due to large average pore size Failure to do so will cause shrinking while transferring, and a distorted pattern of transfer (Table 2). The gel needs to equilibrate for 3-5 minutes in ice cold transfer buffer. Incubate in ice cold transfer buffer for 5 minutes. 
PVDF membranes require careful pre-treatment: Soak it in methanol for 1-2 minutes. This membrane is usually made of nitrocellulose (NC) or polyvinylidene fluoride (PVDF). Use the copper stain if you plan to transfer the separated proteins to a membrane, as the Coomassie stain is not reversible.Īfter completion of the separation of proteins by polyacrylamide gel electrophoresis (PAGE), the next step is to transfer the proteins from the gel to a solid support membrane for further analysis. The visualization of protein at this stage is useful to determine if proteins have migrated uniformly and evenly. Visualization of proteins in gels (Optional): Before protein transfer, we could validating the electrophoresis result by visualization of proteins in gel. Where even loading or transfer have not occurred, the loading control bands can be used to quantify the protein amounts in each lane. They are also useful to check for even transfer from the gel to the membrane across the whole gel. This is important especially when a comparison must be made between the expression levels of a protein in different samples (Table 1). Loading controls are required to check that the lanes in your gel have been evenly loaded with sample. For most runs, it is convenient to reserve at least one separate lane on the gel to run the molecular weight markers. Markers are composed of different proteins of known size and the distances migrated over the time course of the run provide a logarithmic scale by which to estimate the size of unknown proteins. Molecular weight markers are used to define the size of proteins run in a gel. A standard migration buffer (also called running buffer) for PAGE is 1 X Tris-glycine. The gels will be submerged in migration buffer which normally contains SDS, except in native gel electrophoresis. Load 20-40 μg total protein per mini-gel well. Never overfill wells, this could lead to poor data if samples spill into adjacent wells and poorly resolved bands. Take care not to pierce the base of the well with the tip as this will create a distorted band. Use special gel loading tips or a micro-syringe to load the complete sample in a narrow well. In addition, we usually add 0.1mg bromophenol blue as indicator into the sample loading buffer. One potential drawback of this popular system is that disulfide bonds tend to form between cysteine residues at this relatively high pH, although this problem can be alleviated by the addition of a reducing agent to the sample. Laemmli (Tris-glycine) buffering systems are the most commonly used and are comprised of a stacking gel of pH 6.8 and a resolving gel of between pH 8 and 9. Sample Loading of Western Blot Loading buffer: 
Learn more about western blot, please see Previous Section. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
